Nutrition in Pregnancy
Authors
INTRODUCTION
In 1889, Prochownick performed the first serious study of diet in pregnancy.1 He restricted calories, reduced sodium intake, and encouraged minimal weight gain with the goal of reducing infant size in women with contracted pelves to avoid preterm induction of labor. The size of the babies was successfully decreased; however, even with early induction of labor in women with contracted pelves, infant loss was 50% and approximately 5% of the mothers died.
A woman's nutrition intake before and during pregnancy affects not only the growth and development of her baby but can also even have an effect on malformation or mental retardation, if specific deficiency of minerals and vitamins like iodine or folic acid or excessive intake of vitamin A occur in pregnancy. Increased folic acid intake preconceptionally and during pregnancy has been shown to help reduce the development of neural tube defects.2, 3, 4 Abnormalities in intellectual and psychoneuromotor development or endemic cretinism, the most severe form of brain damage caused by iodine deficiency, can be prevented when normal iodine supply is provided to the mother before and during pregnancy, and to the neonate during the critical period of brain development.5 Otherwise excessive vitamin A intake, especially in early pregnancy, has been shown to be teratogenic to the fetus.6
Maternal obesity, a global health problem, raises the risk of several congenital anomalies and infants of obese mothers are at increased risk of birth difficulties, macrosomia, and perinatal death.7
In 1990, the Institute of Medicine (IOM) undertook an extensive review of the data on pregnancy and made recommendations for weight gain, dietary intake, and nutrient supplementation.8 Further in 1992, the IOM published Nutrition during Pregnancy and Lactation: An Implementation Guide, which provides guidelines to counsel pregnant women regarding appropriate dietary intake.9 It is concluded that pregnant women should gain weight based on their pregravid body mass index (BMI), receive counseling on a healthy diet after a routine assessment of dietary intake, to meet recommended weight gain, and be monitored regularly to identify potential problems, with early intervention when indicated. In recent years a series of revisions to the recommended dietary allowances (RDA) for many of the macronutrients (carbohydrates, proteins, and fats) and micronutrients (sodium, potassium, calcium, iron, etc.) have been published and refer to these recommendations as “dietary reference intakes” or DRIs.10, 11, 12, 13, 14 Recommendations for pregnant and lactating women have been included.
The obstetrician is usually the first person from whom the pregnant woman seeks nutrition information. Specific recommendations should be based on nutritional needs and individualized for each woman, taking into account cultural and ethnic background and views regarding diet and health. The impact of pregnancy is not always seen immediately, and may not be seen in the pregnant woman but may manifest in the infant.
There is a growing amount of evidence that maternal nutrition before and during pregnancy can affect the fetus at birth and throughout life.16 However, many scientific questions remain unanswered due to the difficulties in performing high quality scientific research in pregnancy. These challenges include the often unknown critical period, mostly during the first 2 months of pregnancy, when nutrition may impact development; many physiological and interindividual changes that occur over the course of normal pregnancy; and the lack of a good animal model.
Most pregnant women become more interested in their body and in health when pregnancy is confirmed. What they eat becomes more important. They are more concerned about their weight and body image. Many women report that they eat better at the beginning of pregnancy; however, they do not necessarily continue the changes throughout pregnancy, for reasons that are not always apparent.
PRECONCEPTION COUNSELING
What a woman eats before pregnancy is as important as intake during pregnancy. For women of childbearing age, a gynecological visit should include a nutrition assessment. The assessment should include BMI status; chronic disease requiring medical nutrition therapy; drug, alcohol, smoking, vitamin, mineral, and herb intake; history of eating habits; and an assessment of disordered eating or other psychosocial issues that may impact nutritional intake. Folic acid supplements should be initiated for all women of childbearing age. Weight loss should be encouraged for overweight women before contemplating pregnancy as maternal obesity is associated with reduced fertility, an increased risk of a range of fetal structural anomalies, and, during pregnancy, to the increased risk of gestational diabetes, pregnancy induced hypertension, preeclampsia, fetal macrosomia, intrauterine fetal death, cesarean section, and breastfeeding difficulties.7, 17, 18, 19, 20 Women who are underweight should be encouraged to gain weight before pregnancy to decrease the risks of small-for-gestational age (SGA) and low-birth-weight (LBW) infants. The cause of the low BMI should be evaluated; some common causes are disordered eating behaviors, drug use, excess exercise, or gastrointestinal disorders. Women with a history of a chronic illness requiring intervention with medical nutrition therapy should be evaluated to ensure good control of their diet and disease status. Referrals to a registered dietitian should be encouraged for women with over- or underweight.
“FETAL ORIGINS” HYPOTHESIS
The “fetal origins” hypothesis is currently getting much attention and research is underway to test it, but the principle that the nutritional, hormonal, and metabolic environment afforded by the mother may permanently program the structure and physiology of her offspring was established long ago.16, 21, 22 It is a manifestation of the general phenomenon of developmental plasticity, a phenomenon by which one genotype can give rise to a range of different physiological or morphological states in response to different environmental conditions during development.23 It is proposed that during the critical periods of growth, an insult caused by either maternal illness or change in the nutrients supplied to the fetus can interrupt the genetic programming for hormonal axes, causing an increased risk for diseases in adult life.24 A study of a group 15,726 men and women born between 1911 and 1930 in Hertfordshire, England25 showed a reduction in death rates from coronary heart disease as birth weights increased. Similar findings were seen in a study based on 70,000 men and women in the United States,26 and a study of 1200 men in South Wales.27 Birth weight has been inversely related to the development of adult disease such as hypertension, diabetes, and heart disease. One theory holds that hypertension is initiated by the reduced number of cells in key organs, such as the kidney glomeruli found in people who were small at birth which necessarily leads to increased blood flow through each glomerulus, and ultimately to glomerulosclerosis and glomerular loss.28, 29 Animal studies provide support for this theory.30 Another process by which slow fetal growth may be linked to later disease is in the setting of hormones and metabolism. An undernourished baby may establish a 'thrifty' way of handling food, one manifestation of which is insulin resistance, which becomes disadaptive if under-nutrition is followed by over-nutrition.31 In adults with type 2 diabetes, those with the lowest birth weights and who are obese as adults appear to have a decrease in the number of functioning β-cells, which causes a decrease in insulin production in adulthood.32 A third link between low birth weight and later disease is that people who were small at birth have enhanced stress responses.33 Prevention of a substantial proportion of type 2 diabetes, and other disorders linked to insulin resistance, may depend on interventions during development. Improving fetal nutrition remains an important long-term goal.
Other studies have not shown the same relationship to maternal nutrition. Matthews and Yudkin concluded that maternal nutrition in industrialized countries seems to have a small effect on placental weight and birth weights.34 At the Montreal Diet Dispensary, the effects of increasing nutrition and its effects on birth weights have been well established.35 Some suggest that the fetus is not a “perfect parasite” and in fact there is “a complex hormonally orchestrated metabolic interplay between the competing needs of the fetus and the mother”.36 More research is needed to determine the effects of the timing and degree of nutrition insults on the incidence of disease in adult life. In the meantime, woman should be encouraged to eat a balanced diet to meet the weight gain goals and nutritional requirements of pregnancy.
INITIAL PRENATAL VISIT
The initial prenatal visit should include a complete assessment of all the components that may impact on a woman's nutritional status before and during pregnancy including a past obstetric history (prior fetus with neural tube defect, preterm delivery, etc.).
Nutrition assessment requires a holistic approach. Eating behaviors are a personal issue, and how or why a person eats is determined by emotional, physical, and cultural beliefs. The four major components of a nutrition assessment are medical, nutrition, psychosocial, and obstetric histories. The medical history is used to determine preexisting conditions that required medical nutrition therapy like phenylketonuria or diabetes mellitus. If a special diet was required before pregnancy, make an assessment to see if any changes are needed. The past obstetric history can help to identify nutrition-related problems. A previous LBW infant or a large-for-gestational age (LGA) baby can indicate a nutrition problem requiring nutrition intervention. A psychosocial assessment should include information on the patient's feelings about the pregnancy, her support system, emotional status, and body image. Whether the pregnancy was planned or unplanned may determine the motivation a woman has to improve her diet. The woman's support system or lack of support during the pregnancy also affects her eating behavior.
The dietary history should begin with a measured height and weight, pregravid weight, BMI determination (Fig. 1), and weight gain in the current pregnancy. A weight and dieting history can help determine any previous or current eating disorder. Possible signs of an eating disorder include a fear of being weighed or refusing to be weighed, poor weight gain, multiple food aversions, ongoing nausea and vomiting, inappropriate body image, eroded tooth enamel, dehydration, enlarged salivary glands, and low prepregnancy weight. Information regarding vitamin, mineral, and herb use should be obtained, and food resources, such as cooking facilities and food availability, should be assessed. The Women, Infants, and Children's Program (WIC) in th US, which provides milk, eggs, cheese, cereal, and juice throughout pregnancy and lactation, is available to low-income pregnant women based on medical need. Cultural and religious food practices and beliefs should be assessed because they may impact the nutritional intake of a pregnant woman. For example, fasting is required in some religions, and in pregnancy this would not be encouraged. A diet history can be obtained through several different methods. The diet history can be done by a 24-hour recall, where a person relates what they ate during the previous 24 hours. Other methods are a food frequency questionnaire, which lists foods and the frequency per day or per week, and a 2–3-day diet diary, which keeps track of actual intake for the specified time. These methods are often combined to cross check information given. A woman may say she drinks four glasses of milk a day, but further questioning may reveal that she buys only one gallon of milk a week. This would be equivalent to two 8-oz glasses a day, instead of the four reported. It is important to include all foods eaten and beverages consumed. Timing of meals and portions should be ascertained to properly assess the pregnant woman's intake. Questioning a woman about her intended infant feeding practices can begin the process of encouraging breastfeeding.
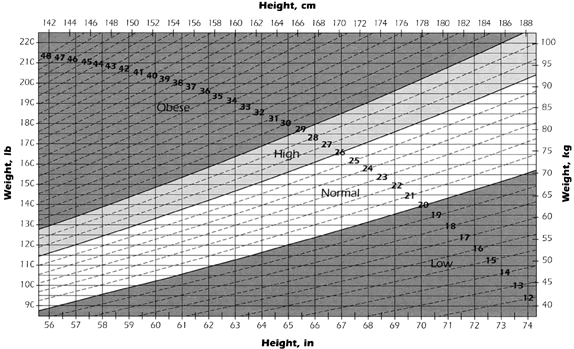
Fig 1. Chart for estimating body mass index (BMI) category and BMI. To find BMI category (e.g., obese), find the point where the woman's height and weight intersect. To estimate BMI, read the bold number on the dashed line that is closest to this point.
The first visit should summarize the nutrition recommendations based on the assessment. Recommendations should include information on weight gain, vitamin and mineral supplementation as needed, promotion of breastfeeding, and treatment of current problems associated with pregnancy, such as nausea, vomiting, heartburn, and constipation. Women found to be at nutritional risk (Table 1) should be referred to a registered dietitian for further assessment and treatment.
Table 1. Nutritional risks in pregnancy
| Review of Past Obstetrical and General History |
| Poor reproductive performance |
| Lactation within 1 year of current pregnancy |
| Short interconceptual periods (<1 year) |
| Bariatric surgery |
| Present Pregnancy |
| Adolescent (<16 year old) |
| Severe nausea and vomiting in early pregnancy |
| Heavy smoker, drug or alcohol use |
| Chronic disease requiring medical nutrition therapy |
| Economically deprived |
| BMI <19.8 or >26 |
| Unwanted pregnancy |
| History of eating disorder or current eating disorder |
| Multiple gestation |
| HIV infection |
| Reduced sun shine exposure |
| Laboratory Findings |
| Hemoglobin <33 g/dl or hematocrit <11% in first or third trimester |
| Hemoglobin <32 g/dl or hematocrit <10.5% in second trimester |
| Ferritin <15 µg/l |
| Clinical Course |
| Individual weight gain not meeting IOM guidelines |
| No fundal height growth in 4 weeks |
NURITIONAL REQUIREMENTS OF PREGNANCY
Calories
The calorie cost of pregnancy is said to be approximately 80,000 calories over the course of pregnancy.37 Calorie requirements should be determined individually based on prepregnancy weight, diet history, height, activity level, singleton or multiple pregnancy, time of gestation, and weight gain goals. The DRIs for energy recommend that pregnant women from 14 to 50 years old consume no additional calories in the first trimester of pregnancy over the level recommended in the USDA "MyPyramid", a new version of the food guide pyramid.38 However, an additional 340 kilocalories per day in the second trimester and 450 kilocalories per day in the third trimester are needed to meet the energy requirements to support the increased maternal metabolic rate and to foster adequate fetal growth.14 Energy intake is adequate when the rate of maternal weight gain meets the IOM guideline.8
Carbohydrates
Carbohydrates should constitute approximately 50% of the diet for most pregnant women. Carbohydrates are important for energy production, and they spare protein from being used as energy. Whole grains, fruits, vegetables, milk, yogurt, beans, and peas contain carbohydrates. The carbohydrate group provides much of the fiber necessary to help prevent constipation and hemorrhoids. At present, the safety of a high-protein, low-carbohydrate diet is unknown. Ketones are produced with low-carbohydrate diets, which may be contraindicated in pregnancy. The RDA for carbohydrates in pregnancy is 175 g/day, up from 130 g/day in nonpregnant women.39
Protein
Protein is required for the growth of maternal and fetal tissue. The effects of a protein deficiency on the outcome of pregnancy are difficult to assess. Most women who have low protein intakes also have a caloric deficit. The pregnant woman should ingest 1.1 g/kg/day protein, which is moderately higher than the 0.8 g/kg/day recommended for nonpregnant adult women and can be met using protein from animal and plant sources.39 Quality as well as quantity of protein should be considered when assessing the protein intake of the pregnant woman. Adolescents and women with multiple gestations have increased protein requirements.
Fat
Fat requirements are unchanged during pregnancy. The American Heart Association guidelines for fat are 30% of total calories per day. The fat intake of a 2000-calorie diet is 60 g/day. Saturated fats should be limited and the majority of fat should be monounsaturated and polyunsaturated.
Long-chain polyunsaturated fatty acids
Humans can synthesize saturated and monounsaturated fatty acids, but they cannot synthesize the n-3 and the n-6 families of polyunsaturated fatty acids (PUFA), they are essential fatty acids. Long-chain polyunsaturated fat acids (LCPUFA) can be consumed as part of the diet or be desaturated and elongated in the human body from the esssential fatty acids. Consequently, the LCPUFA status of the developing fetus depends on that of its mother, as confirmed by the positive relation between maternal PUFA consumption and neonatal PUFA status. This is particularly true for docosahexaenoic acid (DHA) because, on the basis of the current habitual diet, birth spacing appeared to be insufficient for the maternal DHA status to normalize completely.40 DHA accumulates in the fetal brain in the second half of pregnancy and is an important structural component of the retina lipids. The physiological importance of DHA is supported by its active and preferential maternal-fetal transfer, which is mediated by specific fatty acid transfer proteins and membranesbinding proteins that favor placental transport of DHA.41 Because of the decrease in PUFA status during pregnancy, the neonatal PUFA status may not be optimal. This view is supported by the lower neonatal PUFA status after multiple as compared with single births.
A number of studies have demonstrated that intake of fatty fish, fish oils or single cell n-3 LCPUFA oils during pregnancy result sin slightly longer gestation, marginally higher birth weight, and reduced risk of preterm delivery especially in women with low or moderate fish consumption.42, 43, 44, 45 The recurrence of preterm delivery could be reduced by 46% in singletons in a randomized fish oil trial.46 A multinational epidemiological study indicated that higher concentrations of DHA in maternal milk and greater seafood consumption predict a lower prevalence of postpartum depression (PPD). The highest prevalence was found in South Africa (24.5%) and the lowest in Japan (2%).47
Observational studies have shown that the LCPUFA in the maternal diet, in particular DHA, have been associated with better cognitive development, and visual acuity of infants.48 Low maternal seafood intake was associated with increased risk of suboptimal outcomes for verbal intelligence quotient, prosocial behavior, fine motor, communication, and social development between the age of 6 months and 8 years.49
After dissemination of the federal recommendation in January 2001, that pregnant women limit consumption of certain fish because of concerns about mercury contaminations, a reduced consumption of fish, including tuna, dark meat fish, and white meat fish was observed. Because these fish may confer nutritional benefits to mother and infant, the public health implications of these changes remain unclear.50
The Food and Drug Administration (FDA)/Environmental Protection Agency (EPA) published revised guidelines in 2004 for women in childbearing age (Table 2).51
Table 2. FDA/EPA guidelines for women of childbearing age
| Do not eat shark, swordfish, king mackerel, or tilefish because they contain high levels of mercury |
| Eat up to 12 ounces, two average meals, a week of a variety of fish and shellfish that are lower in mercury. Five of the most commonly eaten fish that are low in mercury are shrimp, canned light tuna, salmon, pollock, and catfish. Albacore (white) tuna has more mercury than canned light tuna. When choosing your two meals of fish and shellfish, you may eat up to 6 ounces, one average meal, of albacore tuna per week |
| Check to see if advisories exist concerning the safety of fish caught in local lakes, rivers, and coastal areas. If no advice is available, eat up to 6 ounces per week of fish you catch from local waters, but don't consume any other fish during that week |
| Follow these same recommendations when feeding fish and shellfish to your young child, but serve smaller portions |
Other different international recommendations have been published with the aim of achieving an adequate dietary intake of n-3 LCPUFA by consuming one to two portions of sea fish per week, including fatty fish. Dietary fish should be selected from a wide range of species without undue preference for large predatory fish, which are more likely to be contaminated with methylmercury.52, 53
Vitamin A
Vitamin A is required for its role in vision, cellular growth and differentiation, and immune function. Vitamin A deficiency in HIV-positive pregnant women has been shown to increase maternal-fetal transmission in parts of Africa. Further studies are underway to identify the role of vitamin A in this population.54 Vitamin A is readily available in the food supply of the United States. The best food sources of vitamin A include leafy green and orange vegetables, such as spinach, dark green lettuce, carrots, sweet potato, acorn and butternut squash, cantaloupe, and apricots. Vitamin A deficiency is rare in industrialized countries, but quite common in other parts of the world. In rat studies, vitamin A deficiency showed impaired lung development55 similar to that seen in premature babies with respiratory distress syndrome. These babies are often found to be vitamin A deficient. The vitamin A content of breast milk is associated with the vitamin A status of the mother during the third trimester of pregnancy. Breast milk of the mildly undernourished woman can be vitamin A deficient after the first few weeks of lactation, putting the infant at risk for health problems related to inadequate vitamin A.56, 57 Vitamin A deficiency can develop in women after bariatric surgery if not adaequately substituted.58
A greater concern regarding vitamin A is the teratogenic effect in animal and human studies of excess intake of preformed vitamin A (retinol).6, 59, 60, 61 Beta-carotene, a precursor of vitamin A, does not seem to be teratogenic. In humans it has been found that isoretinoin, a vitamin A analog, has been associated with fetal anomalies involving the central nervous system, and craniofacial, cardiovascular, and thymus malformations.62 Due to the possibility of its teratogenic effects, the World Health Organization (WHO) recommends that vitamin A intake in supplements used before or during pregnancy should not exceed 10,000 IU/day (3000 μg).63 The Teratology Society of the United States recommends total vitamin A intake from food or supplements should not exceed 8000 IU/day (2400 μg).64 In 2001, the Food and Nutrition Board of the National Academy of Sciences recommended a DRI of 770 μg (2500 IU) of vitamin A for pregnant women and an upper tolerable limit of 3000 μg (10,000 IU) per day.65 The IOM recommends avoiding vitamin A supplements, especially in the first trimester.8 Preformed vitamin A is found only in animal products, such as meat, milk, eggs, fish, and chicken. Beef liver has more than three times the amount of preformed vitamin A than is currently recommended. Provitamin A beta-carotene sources includes dark green leafy and orange vegetables and fruits. When vitamin A supplements are prescribed, it is important to check the source of the vitamin A. Some prenatal vitamins do not identify the source of vitamin A as either preformed vitamin A or beta-carotene. Avoid vitamins that do not list the specific type of vitamin A. Women with a low serum retinol concentration and poor intake of vitamin A would require supplementation.
Isoretinoid, a vitamin A analogue, is approved for the treatment of severe acne. Due to its teratogenic effect women of childbearing age should use effective contraceptive methods, and different programs and precautions are instituted. However, pregnancy exposure to isoretinoid continues to occur and even to accumulate, as more than half of the women do not use any form of contraception while on therapy with isoretinoids.66, 67, 68
Folic acid
Inadequate intake of folic acid periconceptionally is associated with an increased risk of neural tube defects. An additional 400 μg of folic acid per day used 3 months before and for the first 12 weeks of pregnancy has been shown to decrease the occurrence of neural tube defects by 70%, odds ratio 0.28 (95% CI 0.13–0.58).2, 3, 4 In addition, the Centers for Disease Control and Prevention recommend that to prevent recurrence of neural tube defects, a folic acid supplement of 4–5 mg/day should be consumed.69 A suspected increase in multiple pregnancies caused by folic acid supplementation could not be confirmed by a large cohort study from China including more than 240,000 pregnant women.70
Folic acid is necessary as a coenzyme in single-carbon transfers from one compound to another. This is important, especially in pregnancy, in the synthesis of several amino acids involved in nucleic acid synthesis. A deficiency of folic acid in pregnancy may impair cell growth and replication and has been associated with poor pregnancy outcome.71 Three sources of folate are currently available. The first source is folate naturally occurring in food. The bioavailability of dietary folate is approximately 50% of synthetic folate.72 In 1998, the FDA required that enriched grains be fortified with folic acid 1.4 μg/g of product.73 These fortified foods are the second source of folic acid. One report suggests the incidence of neural tube defects in the United States has decreased by 19% since the introduction of folate-fortified foods.74 The third source of folic acid is synthetic folate from vitamin supplements. Folic acid in fortified foods and that provided by supplements are absorbed at a rate of 85–100%.75
The recommended DRI for folic acid in pregnancy set by the Food and Nutrition Board of the National Academy of Sciences is 600 μg/day with an upper tolerable intake level of 1000 μg/day.76 A level of 600 μg/day was found to maintain normal folate status. This level can be achieved with a diet high in naturally occurring folate and a supplement of 400 μg/day. Dietary sources of folate include leafy green vegetables, orange juice, strawberries, dried peas, beans, and foods enriched with folate such as whole-grain breads, pasta, rice, and cereals. Women at risk for folic acid deficiency include those with low dietary intake, and those who use medications that are folate antagonists such as trimethoprim, triamterene, carbamazepine, phenytoin, phenobarbital, and primidone.
Most pregnancies are unplanned, and even those that are planned often do not meet the recommendation for folate. At their gynecology visits, women of childbearing age should be advised to take a folic acid supplement and include good food sources of folic acid in their diets.
Vitamin D
Vitamin D is essential for skeleton growth and bone health, and regulates calcium and phospate absorption and metabolism. Nutrition that contains vitamin D is oily fish, egg, fortified margarines, and some breakfast cereals, but the main source is the synthesis following exposure of the skin to sunlight. Vitamin D deficiency can occur when the demand for it exceeds supply, as occurs in a period of rapid growth such as during pregnancy and lactation. Deficiency results in neonatal hypocalcemia, craniotabes, tetania, and severe deficiency of vitamin D results in rickets (in children) and osteomalacia (in children and adults). Two randomized trials with vitamin D supplementation versus placebo involving 232 women were included in a systematic review.77 In one trial the mothers had a higher mean daily weight gain and a lower number of LBW infants. In the other trial the supplemented group had lower birth weights. So far routine supplementation is not recommended and further studies are necessary to determine optimal vitamin D intake for women at risk.78 Special care should be taken regarding women at greatest risk such as women with increased skin pigmentation living in relative sunless regions , women who have limited exposure to sunlight due to religious or cultural practices, women who eat a diet particularly low in vitamin D, and obese women with BMI >30 kg/m².
Iron
Iron deficiency anemia is the most common form of nutritional deficiency. The rate of iron deficiency anemia is high among low-income pregnant women and in developing countries. No improvement in anemia has been seen in this group since the 1970s. Iron deficiency anemia, especially severe and chronic anemia, is associated with a higher incidence of LBW infants and a shorter length of gestation, a higher perinatal mortality and increased maternal mortality and morbidity. Trials have shown that when iron supplementation was provided, the incidence of LBW infants was decreased.79
Iron absorption is increased during pregnancy and depends on maternal iron stores. Heme iron (iron from animal sources) is two to three times more absorbable than nonheme iron, plant-based foods, and iron-fortified foods. Nonheme iron is better absorbed in the presence of heme iron and vitamin C. Absorption is impaired when consumed with phytate (e.g., bran, whole grains), calcium (especially calcium in supplements), tannin in teas, and polyphenol in some vegetables.
The Continuing Survey of Food Intakes by Individuals (1994–1996) (CSFII)80 suggests that one quarter of adolescent girls and women of childbearing age meet the RDA for iron through diet. National Health and Nutrition Examination Survey (NHANES) III data indicate 11% of nonpregnant women had iron deficiency and 3–5% had iron-deficiency anemia.81
The DRI for iron is 27 mg/day. The tolerable upper level of intake is 45 mg/day.65 The American College of Obstetricians and Gynecologists recommended in 2008 that all pregnant women should be screened for anemia during pregnancy at the first prenatal visit and encouraged to use dietary iron from foods with high iron content, and increase foods that enhance iron absorption. Those with iron deficiency anemia should be treated with supplemental iron additional to prenatal vitamin supplementation.82 The cutoff values for anemia in pregnancy are as follows: first trimester, hemoglobin 11 g/dl and hematocrit 33%; second trimester, hemoglobin 10.5 g/dl and hematocrit 32%; and third trimester, hemoglobin 11 g/dl and hematocrit 33%.8
It is unclear whether iron supplementation in nonanemic, well-nourished pregnant women improves birth outcome; data are conflicting.83, 84, 85 A systematic review of 20 randomized controlled trials compared iron supplementation with either placebo or no iron in pregnant women (n = 5552) with normal hemoglobin levels (greater than 10 g/100 ml) at less than 28 weeks of gestation. Routine iron supplementation raised or maintained the serum ferritin level above 10 μg/l and resulted in a substantial reduction in women with a hemoglobin level below 10 or 10.5 g/100 ml in late pregnancy. There was no evidence of any beneficial or harmful effects on maternal or fetal outcomes.86, 87
Women with iron deficiency anemia (first or third trimester hemoglobin [Hb] <11 g/dl or second trimester Hb ≤10.4 g/dl and low serum ferritin) should receive an additional iron supplement of 30–120 mg/day or receive parenteral iron until the anemia is corrected and dietary changes to improve absorption and intake should be discussed. Compliance with iron supplementation is often poor because of associated gastrointestinal side effects. Studies are being undertaken to determine the best way of dosing iron, and have looked at weekly, daily, and multiple daily dosing.
Maternal hemoglobin below 6 g/dl has been associated with reduced amniotic fluid volume, fetal cerebral vasodilation, and nonreassuring fetal heart rate patterns.88 It is prudent to treat severe anemia with red cell transfusion, especially if there are signs suggestive of fetal hypoxemia.
Failure to respond to iron therapy should prompt further investigation and may suggest an incorrect diagnosis, coexisting disease, malabsorption (sometimes caused by the use of enteric-coated tablets or concomitant use of antacids), noncompliance, or blood loss.
Calcium
Calcium is important for bone and tooth formation and maintenance, and is required in nerve transmission and regulation of the heartbeat. During pregnancy, calcium is transferred to the fetus at a rate of approximately 330 mg/day by the third trimester.8 Calcium absorption and urinary excretion of calcium are greater than before pregnancy. These changes are noticeable before fetal calcium demands are actually increased.89 Multiple studies have measured the changes in bone density in pregnant and lactating women. Bone mineral losses are seen by 3 months of postpartum, and by 5 months after menses begin the bone mineral content returns to prepregnant status.90, 91 Lactating adolescents are the one group at risk for permanent bone loss. One study of lactating adolescents showed bone loss in the forearm of those not supplemented with calcium, whereas those supplemented with calcium had little to no bone loss.92 A study of Indian women with typical calcium-deficient diets showed that neonates whose mothers received a calcium supplement had higher bone density than those whose mothers did not receive a supplement.93
Low calcium intake has also been associated with an increased risk of pregnancy-induced hypertension. Bucher conducted a meta-analysis on 14 randomized controlled trials.94 In most of the studies, the usual calcium intakes were below the RDA for calcium. It was found that 375–2000 mg of supplemental calcium had a blood pressure-lowering effect. The study showed calcium supplementation reduced the risk of developing pregnancy-induced hypertension and preeclampsia in women who were normotensive before pregnancy.94 Another study with 4589 healthy nulliparous women at 13–21 weeks of gestation, performed by the Calcium for Preeclampsia Prevention (CPEP) group, found no change in blood pressure in a group of women with usual calcium intakes of 1100 mg/day who were given an additional 2000 mg/day by calcium supplement.95 There were no differences between the two groups in the incidence of preeclampsia (6.9 versus 7.3%), pregnancy-associated hypertension, or adverse fetal outcomes. The differences in Bucher's meta-analysis and the CPEP study may be explained by the latter group having a normal calcium intake.
Meta-analyses of placebo-controlled trials in low-risk women with adequate dietary calcium intake generally showed that calcium supplementation has no benefit in preventing preeclampsia, in contrast to studies in high risk, low calcium intake populations.96 Therefore ,when calcium intakes are within the normal range, additional calcium has no beneficial effect.
The Food and Nutrition Board of the National Academy of Sciences set the adequate intake (AI) for calcium in pregnancy at 1000 mg/day for women age 19–50 years and 1300 mg/day for adolescents under 19 years. The tolerable upper intake level is 2500 mg/day through diet and nutritional supplementation.97 Milk (8 oz), yogurt (8 oz), and cheese (1.5 oz) all provide approximately 300 mg of calcium. Almonds, broccoli, sardines and salmon with bones, soy milk, and calcium-fortified orange juice are also good sources of calcium, although portions vary. Dietary calcium sources should be encouraged. If calcium intake is below the RDA, a calcium supplement should be considered. Calcium carbonate from Tums and other supplements should be taken with a meal. Calcium citrate can be taken with or without meals.
EATING RECOMMENDATIONS
Meal frequency
Women enter pregnancy with many different eating habits. Some women eat one meal a day, whereas others consider themselves to be “grazers” who eat many small meals. There is no one way for every woman to meet her nutritional requirements. An investigation using the data from the Pregnancy, Infection, and Nutrition Study98 concluded that theistudy supports animal model work showing an association between decreased frequency of meals and preterm delivery. A 24-hour fast during Yom Kippur also showed an increased rate of spontaneous deliveries.99 The potential for prolonged periods without food can cause hypoglycemia, which may stimulate neuroendocrine events affecting the fetus.100, 101 Therefore, meal timing should be considered when assessing and counseling a pregnant woman. Most women find meals are best spaced no more than 5 hours apart. If meals must be more than 5 hours apart, a snack including foods from the food guide pyramid is usually recommended. People who often skip meals or go a long time between meals choose more snacks with low nutrient density or overeat at the meals they do eat. A small bedtime snack (approximately 200 calories) is usually encouraged to avoid a long period overnight without eating and to avoid the possibility of ketosis.
Translating nutrition requirements into diet recommendations
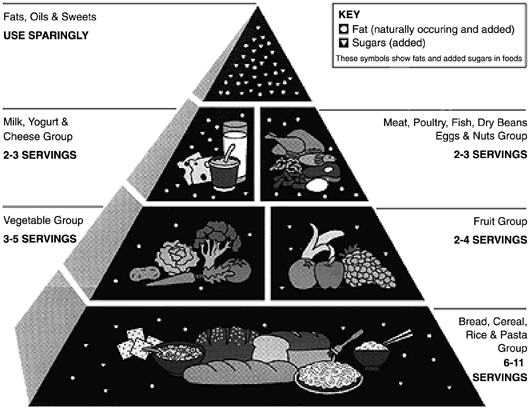
After a nutrition assessment, a review of the food guide pyramid (Fig. 2) can guide the woman in her food choices to better meet the requirements of pregnancy. A variety of foods from each of the groups is encouraged.
Fig 2. Food guide pyramid. For pregnant women, three servings from the milk, yogurt, and cheese group are recommended. (US Department of Agriculture/US Department of Health and Human Services. http://www.nalusda.gov/fnic/Fpyr/pyramid.gif.)
The newer pyramid (Fig. 3) has colored, vertical stripes of varying widths with an outline of a person climbing stairs alongside the pyramid. Each color represents a different food group: orange represents the grains food group; green, the vegetables food group; red, the fruits food group; yellow, oils; blue, the milk food group; and purple, the meat-and-bean food group. The stripes also vary in width to signify the relative proportion of food that should be consumed from each food group on any one day. For example, the wide, orange stripe indicates that most of the food eaten in a day should come from the grain food group, while the narrow, yellow stripe indicates that oils and fats should be consumed sparingly. The outline of a person climbing stairs indicates the increased emphasis on exercise and suggests finding a balance between food and exercise, an initiative that is supported by the USDA.
Fig 3. Food guide pyramid with colored vertical stripes representing different food groups. The person climbing stairs indicates balance between food and exercise.
There is no one perfect food. Foods are used in combinations to provide all of the essential nutrients. Nutrition information provided to each woman should be based on her ethnic and cultural background. The USDA provides a free internet site to help pregnant women choose appropriate foods from a food pyramid ("MyPyramid for Moms") based on their personal characteristics.102 Whole-grain breads and cereals are encouraged, with at least six to 11 servings per day of a variety of grains. Serving sizes are defined as one-half cup rice, pasta, or cereals, or 1 oz or 1 slice of bread or bread product. Women of various ethnic groups may choose tortillas, naan, chapati, or pitta bread as other examples of grains. Grains provide fiber, complex carbohydrates, B vitamins, folic acid, and iron. From the fruit group, three to five servings are recommended each day. Many pregnant women increase their intake of fruit juices because they decrease diet soft drinks. Fruit juices , however, can add many additional calories without the woman being aware. A variety of fresh fruits are encouraged instead of fruit juices. Oranges, strawberries, melons, and other fruits can provide folic acid, vitamin C, and vitamin A in the form of beta-carotene. Vegetables are also recommended, three to five servings per day, from a variety of fresh or frozen vegetables. Vegetables are good sources of fiber, vitamins A and C, and folic acid, and some are high in nonheme iron. Two medium carrots can provide almost all the vitamin A needed for one day in the form of beta-carotene. Dark green leafy vegetables are good sources of vitamin A and folic acid. Dark green lettuce and tomatoes on sandwiches can provide a serving of vegetables. A serving of vegetables is one cup raw or one-half cup cooked vegetables. Vegetables should be added as a side dish to each lunch or dinner or for snacks. Meat and meat substitutes are recommended, with two to three servings of 2–3 oz each per day. Lean meats, chicken, fish, beans, and peas are recommended. Incorporating beans, peas, nuts, and soy proteins can provide adequate protein intake for pregnant women who follow a vegetarian diet. For lacto-ovo vegetarians, eggs and cheese can be added to meet their protein needs. With proper meal planning, vegetarians are able to meet their protein requirements. Most pregnant women eat approximately 75–110 g of protein per day, exceeding their recommended protein requirement for pregnancy. Intake of lunchmeats and high-fat processed meats should be limited. Three or more servings each day are recommended from the milk group, which provides calcium, vitamin D, and protein. A serving includes 8 oz of milk or yogurt or 1.5 oz of low-fat cheese. Chocolate milk, hot chocolate, using milk in soups, adding cheese to sandwiches, or eating yogurt as a snack are ways of increasing calcium intake. Women with lactose intolerance can use lactose-reduced milk. Small, frequent servings of milk products are often better tolerated than larger servings. If calcium intake is less than 1000 mg/day, calcium supplements are recommended. Women who follow the guidelines in the food guide pyramid should be able to meet the requirements for most nutrients.
VITAMIN, MINERAL, AND HERBAL SUPPLEMENTS
The IOM and CDC recommend multivitamin supplements for pregnant women who do not consume an adequate diet.103, 104 Risk factors requiring vitamin and mineral supplementation include multiple gestation, HIV infection, drug or alcohol abuser, heavy smokers, adolescents, complete vegetarians, lactose deficiency, and women with a poor dietary intake with resistance to change.105 Well-nourished women may not need multivitamins to satisfy these daily requirements.106
Folic acid supplementation prevents congenital neural tube defects based on randomized controlled trials, but folic acid containing prenatal multivitamins may also prevent cardiovascular defects, limb defects, cleft palate, urinary tract anomalies, and pediatric cancers. Based on the evidence available today it is impossible to ascertain which component(s) of the prenatal multivitamin supplements are associated with these protective effects. All these available studies were not only randomized but also case-controlled or cohort studies and therefore taking prenatal vitamins may not be causal, but rather a marker of well-being, better socioeconomic class, nutrition, and healthy lifestyle.107, 108
The vitamin supplement should include 30–60 mg iron, 15 mg zinc, 2 mg copper, 250 mg calcium, 400 IU vitamin D, 50 mg vitamin C, 2 mg vitamin B6, 400 μg folic acid, and 2 μg vitamin B12.8 Care should be taken to check the vitamin A source, and beta-carotene rather than retinol or preformed vitamin A should be used.
Herb use
All pregnant women should be questioned about use of herbal supplements. Many herbs are contraindicated in pregnancy because of known adverse effects in pregnant women. Dong quai, foxglove, pokeroot, senna, uva ursi, aloe, chaparral, coltsfoot, St. John's wort, black cohosh, comfrey, and pennyroyal are some herbs that are not recommended in pregnancy because of their potential to either induce abortion or cause central nervous system disturbances or diarrhea. Women often hesitate to tell their health care providers about use of herbs because they fear disapproval. Very few data exist on the effects of herbs in pregnancy on the developing fetus. Currently, there is no regulation of herbs in the United States, and what the label says may not be what the bottle contains. Herbal teas bought in the grocery store can be used in moderation (one to two cups/day). Teas bought in health food stores or from herbalists should be avoided because they are not regulated as food or drug items. At this time, it is best to discourage the use of herbs until more data are available.
NONNUTRITIVE SUBSTANCES
Caffeine
Caffeine is probably the most frequently ingested pharmacologically active substance in the world.109 Caffeine is present in coffee, tea, cola, and chocolate, and the intake has been associated with an increased risk of miscarriage, LBW infants, and a reduction in birth weights. Most of the studies looked at caffeine intakes as low as 150 mg/day and as high as the equivalent of seven cups of coffee a day. In 1990, the IOM subcommittee concluded that the data were insufficient to make any specific recommendations on caffeine intake.5 Nehlig and Derby in 1994 looked at the data from human and animal studies of caffeine intake during pregnancy, and concluded that moderate consumption of coffee during pregnancy should be advised.110 Moderate caffeine intake is considered to be less than 200 mg/day. Using milk in coffee beverages is a common way for women to increase calcium intake. Mexican women often drink their coffee with at least 4–6 oz of milk, which adds more calcium to their diet.
In a large prospective observational study, including 2635 women, the CARE group reported an association between caffeine consumption and the risk for fetal growth restriction. For 100–199 mg caffeine a day the odds ratio was 1.2 (95% CI 0.9–1.6), for 200–299 mg/day it was 1.4 (1.0–2.0), and for over 300 mg/day it was 1.5 (1.1– 2.1). The association of caffeine with fetal growth restriction seemed to be stronger in women with faster caffeine clearance.111 The current body of evidence is limited concerning the maximum daily intake. Sensible advice would be to reduce caffeine intake during pregnancy, but not replace caffeine containing beverages with drinks containing alcohol or soft drinks loaded with sugar. Table 3 lists the caffeine content of some beverages and foods.
Table 3. Caffeine content of foods and beverages
Caffeine Content | Caffeine Equivalents (mg/Serving) |
Coffee, 6-oz cup | 80–200 |
Coffee, brewed drip method (6-oz cup) | 135–195 |
Coffee, percolated (6-oz cup) | 110–160 |
Tea (6-oz cup; depends on steeping time) | 25–70 |
Cola (12-oz glass) | 30–65 |
Hot cocoa (6-oz cup) | 10–20 |
Milk chocolate (1 oz) | 1–15 |
Dark chocolate (1 oz) | 5–35 |
(Adapted from Nutrition and Pregnancy, Northwestern Memorial Hospital)
Alcohol
Alcohol passes freely across the placenta and is a recognized teratogen with effects including a decrease in DNA synthesis, impaired cellular growth and differentiation, changes in placental function, and decrease in glucose and amino acid transfer to the fetus. Spontaneous abortions are twice as likely with moderate alcohol intake (more than 1 oz of alcohol twice a week) and four times more likely with heavy drinking (one to five drinks per day, four times a week). Alcohol is associated with an increase in perinatal mortality, a decrease in birth weights, and an increase in LBW infants and congenital malformations. The fetal alcohol syndrome is characterized by LBW and length, low head circumference, congenital and intellectual anomalies, and certain facial features, and is associated with alcohol intake during pregnancy. Binge drinking has the greatest effect on the fetus; however, some reports have shown adverse effects on neonatal outcomes with as little as one alcoholic beverage every day.112, 113 Growth was impaired in the infants of women who quit by the second trimester, though not to the same extent as in women who continued to drink.114
Many studies do not use biomarkers for measurement of alcohol consumption or do not adjust for confounders like low socioeconomic status and smoking. The sensitivity of the pregnancy to the adverse effects of alcohol varies between women and between the different stages of gestation. The minimum or threshold level at which alcohol begins to pose a significant threat to pregnancy is not known. It is therefore preferable that women avoid the intake of alcohol during pregnancy.
Artificial sweeteners
In 1993, the American Dietetic Association prepared a position statement on the use of nonnutritive sweeteners in pregnancy. They stated that saccharin can cross the placenta and limited use was advised. Aspartame was said to be safe in pregnancy except for women with phenylketonuria (PKU). Studies have been performed showing that intake of aspartame levels at least three times the 99th centile pose no risk to the mother or the developing fetus.115, 116 Of the three components of aspartame, aspartate does not cross the placenta. Phenylalanine does cross the placenta, but the amount is unlikely to cause neurotoxicity. Women without PKU have phenylalanine hydroxylase activity to prevent any substantial or sustained rise in serum phenylalanine. Methanol does cross the placenta, but the amount ingested is very small. Acesulfame K is safe for use in pregnancy.117 Sucralose is another nonnutritive sweetener approved for use in 1998. The FDA reported that sucralose was found to be safe for use by pregnant women.118, 119
Exercise
Regular moderate exercise is encouraged during the pregnancy of a healthy woman.120 The Centers for Disease Control and Prevention and the American College of Sports Medicine (CDC-ACSM) have recommended the accumulation of 30 minutes or more of moderate intensity physical activity on most, and preferably all, days of the week.121 Available data in the Cochrane Library so far are insufficient to infer important risks or benefits for the mother or infant.122 In weight bearing exercise, such as walking, the energy requirement progressively increases with the increase in weight during the course of the pregnancy. A related consideration to nutrition and exercise during pregnancy is adequate carbohydrate intake. Pregnant women use carbohydrates at a greater rate both at rest and during exercise than do nonpregnant women.121 Pregnant women who exercise should be sure to drink plenty of fluids before, during, and after exercise to prevent dehydration. A small snack containing some carbohydrates should be eaten at least 1 hour before exercise to prevent hypoglycemia. A small snack may also be required after exercise based on the length of time and intensity of the workout. For women with poor weight gain, exercise should be limited or more snacks before and after exercise should be encouraged to meet weight gain goals.
WEIGHT GAIN RECOMMENDATIONS
In 1990, the Institute of Medicine of the National Academy of Sciences reviewed the literature and published their report confirming the association previously made regarding pregnancy weight gain and birth weights. They redefined weight gain goals for pregnant women. The goals are based on the woman's prepregnancy BMI. Table 4 shows the recommended weight gains based on the BMI and the rate of weight gain recommended by the IOM.8
Table 4. Weight gain recommendations and rates of weight gain
Weight-for-Height Category | Recommended Total Gain (lb) | Rate of Gain per Week (lb), 2nd and 3rd Trimesters |
Low (BMI <19.8) | 28–40 | 1.25 |
Normal (BMI 19.8–26) | 25–35 | 1.0 |
High (BMI >26–29) | 15–25 | 0.66 |
Obese (BMI >29) | At least 15 | 0.5 |
(Institute of Medicine: Nutrition During Pregnancy. Part I, Weight Gain; Part II, Nutrient Supplements. Washington, DC, National Academy Press, 1990)
Recently weight gain charts have been published including accurate estimated gestational age and differentiation between ethnic populations.123 The weight and BMI centile curves have the advantage in that they consider height during the whole course of pregnancy. Figures 4 and 5 show an additional helpful tool in controlling weight gain in pregnancy.
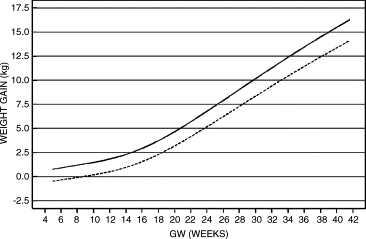
Fig 4. Weight gain 50th centile curves in Caucasians (continuous line) and Asians and Blacks.
Fig 5. BMI 50th centile curves in Caucasians and Blacks (continuous line) and Asians (dashed line).
The RTI International–University of North Carolina at Chapel Hill Evidence-based Practice Center (RTI-UNC EPC) recently systematically reviewed evidence on outcomes of gestational weight gain within or outside the 1990 Institute of Medicine (IOM) guidelines.124 The studies reviewed provided strong evidence for the independent association of pregravid weight status and outcomes. Regarding outcomes of weight gain within or outside 1990 IOM guidelines, moderate to strong evidence suggests an association between weight gain below IOM recommendations and preterm birth, LBW, SGA birth weights, and failure to initiate breastfeeding, and strong evidence for the association between weight gain above IOM recommendations and high birth weight, macrosomia, and LGA birth weights.
Pregnant women and physicians have expressed concern associated with the increased weight gain ranges established by the IOM and postpartum weight retention. The National Maternal and Infant Health Survey examined the association between pregnancy weight gain and weight retention at 10–18 months of postpartum. The women who gained above the IOM guidelines of 16 kg were more likely to have weight gain retention of more than 6 kg. The group of women in or below the normal range for weight gain had retained about the same amount of weight. The National Maternal and Infant Health Survey showed that weight gain retention was less than 1 kg for white women and less than 3 kg for black women gaining weight within the IOM guidelines. Black women are at higher risk for weight gain retention of more than 6 kg at any weight gain. Both black and white women who exceeded the weight gain guidelines of 16 kg were twice as likely to retain more than 9 kg at 10–24 months of postpartum.125 How much of that weight was gained postpartum is unknown.
The rate of weight gain should also be considered when assessing weight gain during the prenatal visit. A Canadian study showed that women who gained more weight in the first 20 weeks of pregnancy were more likely to retain more weight postpartum.126
Weight gain recommendations continue to be an important factor in pregnancy outcome. Total weight gain and the rate of weight gain should be evaluated during each visit especially in the woman in whom nutrition is of concern. There is some evidence that intensive antenatal dietary counseling and support is effective in increasing women’s knowledge about healthy eating and can impact upon eating behaviors.127 A total of 179 healthy pregnant women with normal and overweight early pregnancy BMI were monitored from early pregnancy until 1-year postpartum and compared with a historical control group of 381 women. Low-income women who received the intervention had a significantly reduced risk of excessive gestational weight gain (odds ratio [OR] 0.41, 95% CI 0.20–0.81). Overweight women within this income subgroup were at significantly reduced risk of retaining more than 2.27 kg (OR 0.24, 95% CI 0.07–0.89). However, there was no evidence linking this with improved pregnancy outcomes.
Underweight women
The underweight woman is at increased risk for having a LBW infant. This risk is increased if she has poor weight gain. A woman with a BMI before pregnancy of less than 19.8 is considered in the underweight range based on the IOM weight gain guidelines. Weight gain recommendations for underweight women are 28–40 lb with a rate of gain of approximately 1.2 lb/week.8 These women should have a complete nutrition evaluation to assess normal eating habits, previous or current history of an eating disorder, exercise habits, or any metabolic, gastrointestinal or psychiatric disorders that may impact weight status. An assessment should include history of drug use, food availability, and a diet history to determine caloric intake, meal frequency, portions, and adequacy of nutritional intake. Recommendations should be discussed with the patient to improve her nutrition status during the pregnancy and to meet weight gain guidelines.
Overweight women
The risk of congenital anomalies, including neural tube defects, cardiovascular anomalies, cleft palate, hydrocephaly, and limb reduction anomalies, increases in women with maternal obesity, and infants of obese mothers are at increased risk of birth difficulties, macrosomia, and perinatal death.7 Undiagnosed diabetes and hyperglycemia is one potential explanation for the increased risk.128 A second exlanation is that, maternal obesity has also been associated with nutritional deficiencies, specifically reduced folate levels.129 Deficiencies in other nutrients may underlie the association with other congenital anomalies. Ultrasound scanning is more difficult in obese women, potentially resulting in fewer terminations of pregnancy for fetal anomaly and therefore increased prevalence at birth.130 Women who enter pregnancy with a BMI of more than 27 have an increased risk for diabetes, hypertension, thromboembolic disorders, and urinary tract infections.17 Macrosomia is increased, with a greater increase in cesarean section rates. The recommended weight gain for women entering pregnancy with a BMI of 26–29 is 15–25 lb, with a rate of weight gain of 0.5–0.75 lb/week. For women with a BMI of more than 29, a weight gain of at least 15 lb is recommended, with a rate of weight gain of 0.5 lb/week. The rate of weight gain should also be considered; less than 0.25 lb/week is not recommended for any pregnant women.8
A diet history should be done to assess usual intake and quality of the diet. The goal of nutrition counseling for the overweight patient is to individualize her diet to meet her calorie and nutrient requirements and maintain weight gain according to the IOM guidelines. Exercise (e.g., walking) should be encouraged. Weight loss diets are not recommended in pregnancy. Referral to a dietitian is encouraged for these women.
Normal-weight women
A normal-weight woman has a pregravid BMI of 19.8–26.0. Her weight gain goals are 25–35 lb with a rate of weight gain of 0.5–1 lb/week.8 A diet history on the initial visit should assess for inadequate intakes based on the guidelines in the food guide pyramid or Mypyramid. Weight gain should be monitored at each visit to ensure weight gain within the normal IOM weight gain range. Any excess weight gain or loss should be evaluated and a referral to a dietitian should be made.
Excess weight gain
Excess weight gain can be defined as a weight gain greater than the rate recommended by the IOM guidelines for each BMI group. The IOM recommends a further evaluation of weight gains of more than 6.5 lb per month for all pregnant women. If a woman is gaining too much weight, the possible cause should be assessed and recommendations made to improve the weight gain rates. Some questions to consider are listed in Table 5. Pregnant women often believe that they can gain extra weight because no one will notice. They start eating more sweets and foods they felt were “forbidden” before pregnancy. It may help to remind them that excess weight gain needs to be lost eventually, and to encourage regular exercise. In the first trimester, many women find they are more fatigued and exercise routines become less frequent. The decrease in exercise and increase in intake leads to excess weight gain. Women also decrease their use of artificial sweeteners and increase caloric soft drinks and fruit juices, which can easily lead to excess weight gain. Nutritional assessments of many women have revealed that an increased intake of fruit juices is a main contributor to excess weight. Once the possible causes of excess weight gain are assessed, suggestions for change should be discussed with the woman. A dietitian should be consulted to assess the possible causes and recommend the necessary changes to improve weight gain. After gaining too much weight one month, many women may restrict their intake too much in order to limit weight gain the next month. If a psychosocial issue has been identified, a referral to a social worker or other psychologist should be initiated.
Table 5. Excess weight gain: what to consider
| 1. Check the previous weight gain for an error in weighing or recording. Was the woman weighed with shoes, coat, or heavier clothing? |
| 2. Review her weight gain pattern. If her weight gain was poor on the previous visit, greater weight gain may be expected with improved intake |
| 3. Assess changes in edema |
| 4. Has the woman increased her dietary intake? Is it a holiday season? Is she eating more sweets, soft drinks, or fruit juices? Is she eating in restaurants more often, particularly fast food restaurants? |
| 5. Is there a psychological/social factor for an increased intake. Has she recently left her job, is she on bed rest, is she experiencing depression, stress, or boredom? Some women eat more when stressed or depressed, and some may have a decrease in appetite |
| 6. Has her physical activity decreased due to fatigue or bed rest? |
| 7. Is the patient using drugs such as marijuana, which may increase appetite? |
| 8. Is the patient drinking alcohol? |
| 9. Is the woman eating laundry starch or other pica, which may lead to an increased caloric intake |
(Adapted from Institute of Medicine: Nutrition during pregnancy and lactation: An implementation guide. Washington, DC, National Academy Press, 1992.)
Once the causes for excess weight gain are identified, recommendations should be made and discussed with the patient. If needed additional referrals should be made to a registered dietitian and/or social worker or other mental health professional.
Poor weight gain
Poor weight gain can be defined as weight gain at a lesser rate than that recommended by the IOM guidelines. A woman in the normal BMI range should have a weight gain of 10 lb by 20 weeks of gestation, and a woman in the overweight range should have a weight gain of 5–7 lb by 20 weeks. A woman with a BMI in the underweight range should have a weight gain of approximately 15 lb by 20 weeks. This may vary depending on the number of pounds she is underweight. The IOM defines poor weight gain as less than 2 lb/month for women with BMI in normal weight range, or less than 1 lb/month for women in the high-to-obese weight range.8 A critical review by Carmichael and co-workers looked at the relationship between weight gain and spontaneous preterm delivery. In 11 of the 14 studies, they showed an association between a low rate of pregnancy weight gain and an increased risk of preterm deliveries. The conclusion drawn by the authors was that a rate of weight gain below the lower limits of the normal weight gain range in late pregnancy, recommended by the IOM, increased the risk for preterm births.131 As with excess weight gain, several questions should be asked to assist in evaluating the cause of weight gains below the recommended weight gain ranges for each BMI category (Table 6).
Table 6. Poor weight gain: what to consider
| 1. Assess overall pattern of weight gain. If weight gain was excessive on the previous visit and the woman made adjustments in her diet, her weight gain may be lower on the next visit. If this poor weight gain pattern persists, re-evaluate the diet history and increase the calories as needed |
| 2. Was there a possible error in recording the weight at the previous visit? Was a different scale used? Did the patient weighed with heavier clothing or with shoes on? |
| 3. Assess changes in edema |
| 4. Is the patient experiencing nausea and vomiting, diarrhea, heartburn, or other illness? |
| 5. Has the patient decreased her intake for fear of gaining too much weight? Does she have a history of an eating disorder? |
| 6. Is the patient experiencing early satiety? This is often caused from trying to increase intake of fluids. The increased fluids cause a decrease in appetite and fullness. If this is the case, assess fluid intake. Replace noncaloric fluids with milk, juice, or other calorie- and nutrient-dense fluid |
| 7. Is a psychosocial factor causing a decreased intake? Common causes are an increase in anxiety about the pregnancy, lack of support from significant other or family members, unwanted pregnancy, financial concerns, loss of job |
| 8. Is the woman using street drugs such as cocaine or heroin, which can cause a loss of appetite? |
| 9. Does the woman have money for food? Does she have access to cooking and food storage facilities? |
(Adapted from Institute of Medicine: Nutrition during pregnancy and lactation: An implementation guide. Washington, DC, National Academy Press, 1992)
Common causes of poor weight gain are skipped meals, fear of gaining too much weight, an unwanted, unplanned pregnancy, and lack of support by the partner or family members. Often women say they “just aren't hungry” or “the baby isn't hungry”. This is usually a red flag for some other problem and should be evaluated. A referral for nutrition services is recommended to assess the cause of the poor weight gain, discuss a nutrition plan, and make other recommendations. Referrals for psychosocial services may be made as needed.
SPECIAL CIRCUMSTANCES
Multiple gestation
With the increase in infertility treatments, multiple pregnancies are more common. Women with multiples present many nutrition-related problems, including increased needs for calories, protein, vitamins, and minerals. Weight gain recommendations are increased. The IOM guideline for a twin gestation is 35–45 lb with a weekly weight gain of approximately 1.5 lb/week. In triplet pregnancy, a gain of approximately 1.5 lb per week beginning early in pregnancy yields a 50-lb total gain.132
Women with multiples need individualized nutrition counseling to help them meet their pregnancy requirements. Many women are told to increase their fluid intake, which causes early satiety and decreased intake at meals. This often results in poor weight gain. Data have shown that when twin birth weights are similar to that of a singleton birth weight, perinatal mortality declines.8 A study by Luke and colleagues concluded that maternal weight gain and pregravid BMI were predictors of fetal birth weight in twin gestations. They also found that women who were seen by a registered dietitian had the highest weight gains and the lowest incidence of babies weighing less than 1500 g.133
Women pregnant with twins should be encouraged to eat small frequent meals accompanied by small amounts of fluids, and to spread between-meal fluid consumption throughout the day. Calcium intake should be increased, though no exact recommendations have been made for multiples. General nutrition guidelines include eating regular meals, using snacks to add foods missed at regular mealtimes, and adding fluids between meals. If weight gain is poor, fluids with caloric value should be used in place of water. If weight gain is excessive, assess portion sizes, edema, and fluid intake. Make necessary changes in the diet to meet the woman's goals.
A 2002 Position of the American Dietetic Association recommends that women pregnant with multiples “consume 3 or more servings from the milk, yogurt, and cheese group; the meat, poultry, fish, dry beans, eggs, and nuts group; and the vegetable group . . . In addition, 2 or more servings from the fruit group and 6 or more servings from the bread, cereal, rice, and pasta group should likely be advised”.134
The Institute of Medicine suggests a 30-mg iron supplement with low-to-moderate doses of zinc, copper, calcium, vitamin B-6, folate, and vitamin C and D after 12 weeks.135 The risk of preterm delivery, LBW, and fetal growth restriction may be elevated by low concentrations of folate.136 The Institute of Medicine recommends supplementation with 600 μg folate daily throughout pregnancy. Assessment of iron status is important for optimal perinatal care due to the high incidence of anemia in multiple pregnancies. The need for calcium and vitamin D seems higher in multifetal than in singleton gestations.137 It is recommended that women with multiples take a calcium supplement and consume two to three servings of dairy food each day.
Adolescent pregnancy
Adolescents experience many physical and psychosocial changes. Their nutritional needs are increased at the time of peak growth. Increased growth increases the demand for iron, zinc, folate, calcium, protein, and calories.138 However, the adolescents most likely to become pregnant are often those with inadequate nutritional status and unfavorable socioeconomic background. Pregnancy further increases nutrient requirements, as previously discussed. Research indicates that pregnant adolescents may be competing for nutrients with their fetus.139
Due to peer pressure, teens often are more concerned about body image. They may skip meals and eat more foods with low nutrient density; however, intake of fast foods with high fat and saturated fat content often increases. Soft drinks replace milk products, so calcium intakes are below the RDA.
Adolescents, especially those younger than 15 years of age, are at higher risk for medical complications of pregnancy than adult mothers. These problems include abnormal maternal weight gain, pregnancy-induced hypertension, anemia, lung disease, and renal disease. Adverse pregnancy outcomes, such as LBW (less than 2500 g) and preterm delivery (less than 37 weeks' gestation), are more than twice as common in adolescent as in adult pregnancies, and the neonatal mortality rate is almost three times higher.140
Weight gain recommendations are the same as for pregnant adults, although weight gain at the higher end of the range is encouraged for adolescents. Teens who gained less than 20 lb had babies weighing 400 g less than teens who gained more than 20 lb. The incidence of LBW babies was 13% for the adolescents gaining less than 20 lb and 1% for those gaining more.141 Pregnant adolescents should be advised to choose foods from the food guide pyramid, and fruits and vegetables should be encouraged. For the adolescent, 1300 mg of calcium are recommended, which would include four servings of milk per day. Fast foods and snack foods should be incorporated into their diets to improve adherence. Multivitamins with minerals are recommended on an individual basis.
There are a number of studies of maternal intake in adolescent mothers, all of which show that teenage mothers may not meet all their nutrient needs.142 One study showed that dietary sugar intake had an adverse effect on pregnancy outcome.143 The population studied were 594 adolescent mothers aged 13–19 years, of whom 61% were black, 30% were Hispanic (Puerto Rican), and 9% were white. High sugar consumption was 206 g sugar/day (n = 60). Adolescents consuming high sugar diets are at twice (9 vs. 17%) the risk of delivering SGA infants. In a representative sample of 300 adolescent mothers, Scholl et al. showed that those who had inadequate weight gain ingested on average less energy (1878 vs. 2232 kcal/day) and they also ingested less protein.144 Associated with the inadequate weight gain was an average decrease in birth weight of 180 g and an increased prevalence of LBW. In light of the increased dietary requirements nutrition services have been available in larger cities in the USA since at least the 1970s, mostly through the Women, Infants and Children (WIC) Special Nutrition Program.145 Nutrition intervention studies have been shown to improve pregnancy outcome in adolescent mothers.146
Pica
Pica is the abnormal craving and ingestion of nonfood substances with little or no nutritional value. The most common forms of pica are laundry starch and clay or dirt. Pica can also include substances like ashes, ice scraped from the walls of the freezer, kitchen cleansers, mothballs, and many others. Many women in different racial, ethnic, and social groups have reported pica.
Pica can result in poor weight gain due to a reported lack of appetite. This is most often associated with eating ice. The ingestion of clay and laundry starch can cause a decrease in nutrient-dense foods even though weight gain may appear normal or even excessive. A 1-lb box of laundry starch contains approximately 1600 calories. Adverse effects of pica include fecal impaction, constipation, decreased absorption of nutrients (e.g., a decrease of iron absorption when clay is consumed), contraction of fillings in teeth (causing an increase in tooth decay), or hemolytic anemia in the mother or fetus.
It is important to ask questions about pica in a nonjudgmental manner. Examples of nonjudgmental questions that may elicit information: “Is there anything you like to eat now that you didn't eat before you got pregnant?” or “Do you crave anything special?”
Some recommendations for women with pica include replacing laundry starch with nonfat milk powder, eating frozen fruit pops or juices instead of ice, or sucking on hard candies, especially lemon candies or mints. Explaining the possible harmful effects of pica on the fetus and suggesting the alternative behaviors may help.
Listeria
Pregnant women are at an increased risk for developing listeria, which can cause an increase in miscarriage, fetal death, or severe illness in the mother. Pregnant women are advised to avoid hot dogs or lunch meat unless heated until steaming hot; soft cheeses such as brie, feta, Camembert, blue cheese, and Mexican cheeses like “queso blanco fresco”; fresh pates or meat spreads; smoked fish and seafood like salmon, whitefish, trout, cod, tuna, or mackerel; raw unpasteurized milk or foods containing raw milk.147
COMMON DISCOMFORTS OF PREGNANCY
Nausea and vomiting
Hormonal changes are thought to cause simple nausea and vomiting of pregnancy. Hormonal increases may initiate nausea through the following mechanism:
The gastrointestinal tract is sensitive to the sudden increase in hormones.
Hypoglycemia may occur due to the physiologic mechanisms set up to shunt glucose to the fetus.
Gastric motility is decreased.
Symptoms of nausea may vary. Some women complain of nausea occurring only in the morning. Others experience nausea throughout the day. Many working women find nausea worse in the late afternoon, when they go the longest without eating. Symptoms may be mild or severe.
In simple cases of nausea, symptoms appear around the 6th week of pregnancy and abate by the 12th week. Excessive vomiting can cause weight loss due to an inadequate intake of calories, protein, and minerals. Eating small frequent meals, including a bedtime snack, and drinking fluids between rather than with meals usually improve the nausea and vomiting. Usually, no particular foods can help decrease or increase the nausea. Pregnant women report different food tolerances. When nausea occurs, women commonly eat less frequently in an attempt to avoid vomiting; however, this often seems to exacerbate the vomiting. Timing of meals and snacks ,and avoiding the feeling of hunger seem to be most important in improving the nausea and vomiting. Some women eat every 2 or 3 hours, whereas others may have to eat more frequently. Most women can go no more than 2.5 hours without eating before the nausea returns. Cooking meals should be avoided if the woman finds that smelling food while cooking increases the nausea. Sandwiches are a good alternative to a cooked meal because of shorter exposure to the food during preparation. It may be recommended to cook when the nausea is decreased, and to reheat the food at meal times. Milk in the morning seems to be difficult for many women, but may be no problem later in the day. Women with nausea and vomiting, particularly those losing weight, should be referred to a registered dietitian for individualized counseling. An example of small frequent meals should be reviewed with the patient (Table 7).
Table 7. Menu suggestions for nausea and vomiting
| Breakfast (within 1 h of waking up) |
| 1 slice toast |
| 1 oz cheese or 1 egg or 2 tablespoons peanut butter |
| 2–2.5 h later |
| 1 slice toast or ½ bagel |
| 1 oz cheese or 1 egg or 2 tablespoons peanut butter or 1 oz turkey or ham |
| Lunch (2–2.5 h later) |
| Half sandwich (meat and cheese) |
| 1 piece of fruit |
| 2–2.5 h later |
| Half sandwich |
| Yogurt |
| Dinner (2–2.5 h later) |
| 2 oz meat, chicken |
| Potato or rice or pasta |
| Vegetables (raw or cooked) |
| Fruit |
| 2–2.5 h later |
| Cereal |
| Milk |
| Fruit |
A bedtime snack is very important. All meals can be changed to suit the patient's tastes. Dinner may be a sandwich or other food that does not require much preparation. Timing of meals and fluid is very important. Fluids should be consumed 30–60 min after meals.
Hyperemesis gravidarum develops in about 2% of all pregnancies. This is persistent vomiting that often requires hospitalization, intravenous fluids, and electrolyte replacement. Once hydrated, the patient should be encouraged to begin a regular diet with small frequent meals. Most women find the clear liquid diet not easy to tolerate, and do better with solid food. If the vomiting continues, tube feedings or parenteral nutrition may be used.
Constipation
Progesterone is thought to slow the peristaltic action of the smooth muscles in the bowel. This, combined with the pressure of the enlarging uterus on the bowel, slows motility and causes constipation. Iron supplementation may also contribute to constipation. Increasing fiber intake can minimize constipation. Whole-grain breads and cereals, fresh and dried fruits and vegetables (especially those with skins), and an increased fluid intake can help alleviate constipation. Increased activity is also encouraged.
Heartburn
Heartburn is common in pregnancy, especially in the third trimester. Heartburn results from the pressure of the enlarging uterus on the stomach and intestines. Progesterone decreases the integrity of the cardiac sphincter, allowing a reflux of food and acid. Antacids can be used if needed. Dietary changes can help prevent heartburn. Recommended changes include small frequent meals, fluids between meals, avoiding tight clothing, eating a snack 1–1.5 hours before bed time, elevating the head of the bed when sleeping, and avoiding high-fat foods and foods that cause heartburn for the individual woman.
REFERENCES
Simpson JW, Lawless RW, Mitchell AC: Responsibility of the obstetrician to the fetus. Obstet Gynecol 45: 481, 1975 |
|
Czeizel AC, Duda I: Prevention of the first occurrence of neural tube defects by periconceptional vitamin supplementation. N Engl J Med 327: 1832, 1992 |
|
MRC Vitamin Research Study Research Group: Prevention of neural tube defects: Results of the Medical Research Council vitamin study. Lancet 338:131, 1991 |
|
Lumley J, Watson L, Watson M, Bower C: Periconceptional supplementation with folate and/or multivitamins for preventing neural tube defects. Cochrane Database Syst Rev 2001;(3):CD001056 |
|
Delange F. Iodine deficiency as a cause of brain damage. Postgrad Med J 2001 Apr;77(906):217-20. Review |
|
Lammer EJ, Chen DT, Hoar RM et al: Retinoic acid embryopathy. N Engl J Med 313: 837, 1985 |
|
Stothard KJ, Tennant PW, Bell R, Rankin J. Maternal overweight and obesity and the risk of congenital anomalies: a systematic review and meta-analysis. JAMA 2009 Feb 11;301(6):636-50 |
|
Institute of Medicine, Committee on Nutritional Status During Pregnancy and Lactation, Food and Nutrition Board: Nutrition During Pregnancy. Part I, Weight Gain; Part II, Nutrient Supplements. Washington, DC, National Academy Press, 1990 |
|
Institute of Medicine. 1992. Nutrition during pregnancy and lactation: An implementation guide. Washington, DC: The National Academies Press |
|
Institute of Medicine. 1997. Dietary Reference Intakes for calcium, phosphorus, magnesium, vitamin D and fluoride. Washington, DC: The National Academies Press. |
|
Institute of Medicine. 1998. Dietary Reference Intakes for thiamine, riboflavin, niacin, vitamin B6, vitamin B12, pantothenic acid, biotin, and choline. Washington, DC: The National Academies Press. |
|
Institute of Medicine. 2000. Dietary Reference Intakes for vitamin C, vitamin E, selenium, and carotenoids. Washington, DC: The National Academies Press. |
|
Institute of Medicine. 2001. Dietary Reference Intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, manganese, molybydenium, nickel, silicon, vanadium, and zinc. Washington, DC: The National Academies Press. |
|
Institute of Medicine. 2005. Dietary Reference Intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein and amino acids (macronutrients). Washington, DC: The National Academies Press. |
|
US Department of Agriculture [USDA]. 2005. Dietary guidelines for Americans 2005. Retrieved March 21, 2006, from http://www.healthierus.gov/dietaryguidelines/. |
|
Godfrey KM, Barker DJP: Fetal nutrition and adult disease. Am J Clin Nutr 7 (suppl): 1344S, 2000 |
|
Garbaciak JA, Richter MD, Miller S et al: Maternal weight and pregnancy complications. Am J Obstet Gynecol 152: 238, 1985 |
|
Bhattacharya S, Campbell DM, Liston WA, Bhattacharya S. Effect of Body Mass Index on pregnancy outcomes in nulliparous women delivering singleton babies. BMC Public Health 2007 Jul 24;7:168 |
|
Weiss JL, Malone FD, Emig D, Ball RH, Nyberg DA, Comstock CH, Saade G, Eddleman K, Carter SM, Craigo SD, Carr SR, D'Alton ME; FASTER Research Consortium. Obesity, obstetric complications and cesarean delivery rate--a population-based screening study. Am J Obstet Gynecol 2004 Apr;190(4):1091-7 |
|
Li R, Jewell S, Grummer-Strawn L. Maternal obesity and breast-feeding practices. Am J Clin Nutr 2003 Apr;77(4):931-6 |
|
Barker DJP: Mothers and Babies and Health in Later Life. Edinburgh, Harcourt, Brace, 1998 |
|
Barker DJP. Fetal origins of coronary heart disease. BMJ 1995; 311: 171–4 |
|
Barker DJ. The developmental origins of insulin resistance. Horm Res. 2005;64 Suppl 3:2-7. Epub 2006 Jan 20 |
|
Lucas A: Programming by early nutrition in man. In Bock GR, Whelan J (eds): The Childhood Environment and Adult Disease, pp 38–55. Chichester, United Kingdom, John Wiley, 1991 |
|
Osmond C, Barker DJP, Winter PD et al: Early growth and death from cardiovascular disease in women. BMJ 307: 1519, 1993 |
|
Rich-Edwards JW, Stampfer MJ, Manson JAE et al: Birth weight and the risk of cardiovascular disease in a cohort of women followed up since 1976. BMJ 315: 396, 1997 |
|
Frankel S, Elwood P, Sweetnam P et al: Birth weight, body mass index in middle age and incident coronary heart disease. Lancet 348: 1478, 1996 |
|
Brenner BM, Chertow GM: Congenital oligonephropathy: An inborn cause of adult hypertension and progressive renal injury? Curr Opin Nephrol Hypertens 1993;2:691-695 |
|
Ingelfinger JR: Is microanatomy destiny? N Engl J Med 2003;348:99-100 |
|
Moritz KM, Dodic M, Wintour EM: Kidney development and the fetal programming of adult disease. Bioessays 2003;25:212-220 |
|
Phillips DIW: Insulin resistance as a programmed response to fetal undernutrition. Diabetologia 1996;39:1119-1122 |
|
Phillips DIW, Hirst S, Clark PMS et al: Fetal growth and insulin secretion in adult life. Diabetologia 37: 592, 1994 |
|
Barker DJP, Forsen T, Uutela A, Osmond C, Eriksson JG: Small size at birth and resilience to the effects of poor living conditions in adult life: longitudinal study. BMJ 2001;323:1273-1276 |
|
Matthews F, Yudkin P: Influence of maternal nutrition on outcome of pregnancy: Prospective cohort study. BMJ 319 (7206): 339, 1999 |
|
Dubois S, Coulombe C, Pencharz PB et al: Ability of the Higgins Nutrition Intervention Program to improve adolescent pregnancy outcome. J Am Diet Assoc 97 (8): 871, 1997 |
|
Prentice AM, Goldberg GR: Energy adaptations in human pregnancy: Limits and long-term consequences. Am J Clin Nutr 71 (suppl): 1226S, 2000 |
|
Hytten F, Chamberlain G: Alimentary function in the physiology of human pregnancy. Clinical Physiology in Obstetrics, pp 151–175. Oxford, Blackwell, 1980 |
|
US Department of Agriculture [USDA]. 2005b. MyPyramid.gov. Retrieved March 21, 2006, from http://www.mypyramid.gov/. |
|
Institute of Medicine. Dietary Reference Intakes: The Essential Guide to Nutrient Requirements. Washington, DC. National Academies Press, 2006 |
|
Hornstra G. Essential fatty acids in mothers and their neonates. Am J Clin Nutr 2000 May;71(5 Suppl):1262S-9S |
|
Koletzko B, Larque E, Demmelmair H. Placental transfer of long-chain polyunsaturated fatty acids (LC-PUFA). J Perinat Med. 2007;35:S5–11. |
|
Horvath A, Koletzko B, Szajewska H. Effect of supplementation of women in high-risk pregnancies with long-chain polyunsaturated fatty acids on pregnancy outcomes and growth measures at birth: a meta-analysis of randomized controlled trials. Br J Nutr. 2007;98:253–9. |
|
Makrides M, Duley L, Olsen SF. Marine oil, and other prostaglandin precursor, supplementation for pregnancy uncomplicated by pre-eclampsia or intrauterine growth restriction. Cochrane Database Syst Rev. 2006;3 |
|
Szajewska H, Horvath A, Koletzko B. Effect of n-3 long chain polyunsaturated fatty acid supplementation of women with low-risk pregnancies on pregnancy outcomes and growth measures at birth: a meta-analysis of randomized controlled trials. Am J Clin Nutr. 2006;83:1337. |
|
Olsen SF, Østerdal ML, Salvig JD, Weber T, Tabor A, Secher NJ. Duration of pregnancy in relation to fish oil supplementation and habitual fish intake: a randomised clinical trial with fish oil. Eur J Clin Nutr 2007 Aug;61(8):976-85. Epub 2007 Feb 7. |
|
Fish Oil Trials In Pregnancy (FOTIP) Team. Olsen SF, Secher NJ, Tabor A, Weber T, Walker JJ, Gluud C. Randomised clinical trials of fish oil supplementation in high risk pregnancies. BJOG 2000 Mar;107(3):382-95 |
|
Hibbeln JR. Seafood consumption, the DHA content of mothers’ milk and prevalence rates of postpartum depression: a cross-national, ecological analysis. J Affect Disord. 2002;69:15–29. |
|
Uauy R, Peirano P, Hoffman D et al: Role of essential fatty acids in the function of the developing nervous system. Lipids 31: 167, 1996 |
|
Hibbeln JR, Davis JM, Steer C, Emmett P, Rogers I, Williams C, Golding J. Maternal seafood consumption in pregnancy and neurodevelopmental outcomes in childhood (ALSPAC study): an observational cohort study. Lancet 2007 Feb 17;369(9561):578-85 |
|
Oken E, Kleinman KP, Berland WE, Simon SR, Rich-Edwards JW, Gillman MW. Decline in fish consumption among pregnant women after a national mercury advisory. Obstet Gynecol 2003 Aug;102(2):346-51 |
|
http://www.fda.gov/fdac/features/2004/304_fish.html |
|
Koletzko B, Lien E, Agostoni C, Böhles H, Campoy C, Cetin I, Decsi T, Dudenhausen JW, Dupont C, Forsyth S, Hoesli I, Holzgreve W, Lapillonne A, Putet G, Secher NJ, Symonds M, Szajewska H, Willatts P, Uauy R; World Association of Perinatal Medicine Dietary Guidelines Working Group. The roles of long-chain polyunsaturated fatty acids in pregnancy, lactation and infancy: review of current knowledge and consensus recommendations. J Perinat Med 2008;36(1):5-14 |
|
http://www.eatwell.gov.uk/agesandstages/pregnancy/ |
|
Semba RD: Overview of the potential role of vitamin A in mother-to-child transmission of HIV-I. Acta Paediatr 421 (suppl): 107, 1997 |
|
Wellik DM, Norback DH, Deluca HF: Retinol is specifically required during midgestation for neonatal survival. Am J Physiol 272: E25, 1997 |
|
Humphrey JH, West KP, Sommer A: Vitamin A deficiency and attributable mortality among under 5 year olds. WHO Bulletin OMS 70: 225, 1992 |
|
West KP, Howard GR, Sommer A: Vitamin A and infection: Public and health implications. Annual Rev Nutr 9: 63, 1989 |
|
Guelinckx I, Devlieger R, Vansant G. Reproductive outcome after bariatric surgery: a critical review. Hum Reprod Update 2009 Mar-Apr;15(2):189-201. Epub 2009 Jan 8 |
|
Hathcock JN, Hattan GD, Jenkin MY et al: Evaluation of vitamin A toxicity. Am J Clin Nutr 52: 183, 1990 |
|
Werler MM, Lammer EJ, Rosenberg L et al: Maternal vitamin A supplementation in relation to selected birth defects. Teratology 42: 497, 1990 |
|
Mills JL, Simpson JL, Cunningham GC et al: Vitamin A and birth defects. Am J Obstet Gynecol 177: 31, 1996 |
|
Benke PJ: The isotretinoin teratogen syndrome. JAMA 251: 3267, 1984 |
|
World Health Organization: Safe vitamin A dosage during pregnancy and lactation. Recommendations and report from a consultation. Micronutrient series (WHO.NUT/964). Geneva, World Health Organization, 1998 |
|
Teratology Society: Recommendations for vitamin A use during pregnancy (Position paper). Teratology 35:269, 1987 |
|
Food and Nutrition Board, Institute of Medicine: Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. Washington, DC, National Academy Press, 2001. Available at www.nap.edu. |
|
Berard A, Azoulay L,Koren G, Blais L, Perreault S, Oraichi D. Isotretinoin, pregnancies, abortions and birth defects: a population-based perspective. Br J Clin Pharmacol 2007:196–205. |
|
Abroms L, Maibach E, Lyon-Daniel K, Feldman SR. What is the best approach to reducing birth defects associated with isotretinoin? PLoS Med 2006:e483. |
|
Garcia-Bournissen F, Tsur L, Goldstein LH, Staroselsky A, Avner M, Asrar F, Berkovitch M, Straface G, Koren G, De Santis M. Fetal exposure to isotretinoin-an international problem. Reprod Toxicol 2008 Jan;25(1):124-8. Epub 2007 Oct 25 |
|
Recommendations for the use of folic acid to reduce the number of cases of spina bifida and other neural tube defects. MMWR Morb Mortal Wkly Rep 41:1, 1992. Available at http://www.cdc.gov/epo/mmwr/preview/mmwrthml/ 00019479.htm. |
|
Li Z, Gindler J, Wang H, Berry RJ, Li S, Correa A, et al. Folic acid supplements during early pregnancy and likelihood of multiple births: a population-based cohort study. Lancet 2003;361:380–4. |
|
Scholl TO, Hediger ML, Schall JI et al: Dietary and serum folate: Their influence on the outcome of pregnancy. Am J Clin Nutr 63: 520, 1996 |
|
Sauberlich HE, Kretsch MJ, Skala JH HL et al: Folate requirement and metabolism in non-pregnant women. Am J Clin Nutr 46: 1016, 1987 |
|
Schaller DR, Olson BH: A food industry perspective on folic acid fortification. J Nutr 126 (suppl): 761s, 1996 |
|
Cuskelly GJ, McNulty H, Scott JM: Effect of increasing dietary folate on red cell folate: Implications for prevention of neural tube defects. Lancet 347: 657, 1996 |
|
Honein MA, Paulozzi CJ, Matthew TJ et al: Impact on folic acid fortification of the US food supply on the occurrence of neural tube defects. JAMA 285 (23): 2981, 2001 |
|
Food and Nutrition Board, Institute of Medicine: Dietary reference intakes for thiamin, riboflavin, niacin, vitamin B6, folate, vitamin B12, pantothenic acid, biotin, and choline. Washington, DC, National Academy Press, 1998. Available at www.nap.edu. |
|
Mahomed K, Gulmezoglu, A. M. Vitamin D supplementation in pregnancy. Cochrane Database of Syst Rev 2000;(2):CD000228. |
|
Hollis BW, Wagner CL.Assessment of dietary vitamin D requirements during pregnancy and lactation. Am J Clin Nutr 2004 May;79(5):717-26. Review |
|
Hemminiki E, Starfield B: Routine administration of iron and vitamins during pregnancy: Review of controlled clinical trials. Br J Obstet Gynaecol 85: 404, 1978 |
|
US Department of Agriculture, Agricultural Research Service: Data tables: Results from USDA's 1994-96 Continuing Survey of Food Intakes by Individuals and 1994-96 Diet and Health Knowledge Survey (online). Riverdale, MD, US Department of Agriculture, Agricultural Research Service, Beltsville Human Nutrition Research Center, December 1997. Available at http://www.barc.suda.gov/bhnrc/foodsurvey/home.htm. |
|
Looker AC, Dallman PR, Carroll MD et al: Prevalence of iron deficiency in the United States. JAMA 277 (12): 973, 1997 |
|
ACOG Practice Bulletin No. 95: anemia in pregnancy. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2008 Jul;112(1):201-7 |
|
Siega-Riz AM; Hartzema AG; Turnbull C; Thorp J; McDonald T; Cogswell ME. The effects of prophylactic iron given in prenatal supplements on iron status and birth outcomes: a randomized controlled trial. Am J Obstet Gynecol. 2006 Feb;194(2):512-9 |
|
Cogswell ME; Parvanta I; Ickes L; Yip R; Brittenham GM. Iron supplementation during pregnancy, anemia, and birth weight: a randomized controlled trial. Am J Clin Nutr 2003 Oct;78(4):773-81 |
|
Makrides M; Crowther CA; Gibson RA; Gibson RS; Skeaff CM. Efficacy and tolerability of low-dose iron supplements during pregnancy: a randomized controlled trial. Am J Clin Nutr 2003;78(1):145-53 |
|
Mahomed K. Iron and folate supplementation in pregnancy. Cochrane Database Syst Rev 2001;(2):CD001135. Review. Update in: Cochrane Database Syst Rev. 2006;(3):CD001135. |
|
Pena-Rosas J; Viteri F. Effects of routine oral iron supplementation with or without folic acid for women during pregnancy. Cochrane Database Syst Rev. 2006 Jul 19;3:CD004736. |
|
Carles G; Tobal N; Raynal P; Herault S; Beucher G; Marret H; Arbeille P. Doppler assessment of the fetal cerebral hemodynamic response to moderate or severe maternal anemia. Am J Obstet Gynecol 2003 Mar;188(3):794-9 |
|
Heany RP, Skillman TG: Calcium metabolism in normal human pregnancy. J Clin Endocrinol 33: 661, 1971 |
|
Ritchie LD, Fung EB, Halloran BP et al: A longitudinal study of calcium homeostasis during human pregnancy and lactation and after resumption of menses. Am J Clin Nutr 67: 693, 1998 |
|
Laskey MA, Prentice A, Hanratty LA et al: Bone changes after 3 months of lactation: Influences of calcium intake, breast milk output and Vitamin D receptor genotype. Am J Clin Nutr 67: 685, 1998 |
|
Chan GM, McMurry M, Westover K et al: Effects of increased dietary calcium intake upon the calcium and bone mineral status of lactating adolescents. Am J Clin Nutr 46: 319, 1987 |
|
Raman L, Rajalakshmi K, Krishnamachari KA et al: Effect of calcium supplementation on undernourished mothers during pregnancy on the bone density of the neonates. Am J Clin Nutr 31: 466, 1978 |
|
Bucher HC, Guyatt GH, Cook RJ, Hatala R, Cook DJ, Lang JD, Hunt D. Effect of calcium supplementation on pregnancy-induced hypertension and preeclampsia: a meta-analysis of randomized controlled trials. JAMA. 1996 Apr 10;275(14):1113-7. Erratum in: JAMA 1996 Nov 6;276(17):1388 |
|
Levine R, Esterlitz J, Raymond E et al: Trial of calcium for preeclampsia prevention (CPEP): Rationale, design and methods. Control Clin Trials 22: 425, 1996 |
|
Atallah, AN, Hofmeyr, GJ, Duley, L. Calcium supplementation during pregnancy for preventing hypertensive disorders and related problems (Cochrane review). Cochrane Database Syst Rev. 2002; 1: CD001059. Update in: Cochrane Database Syst Rev. 2006;3:CD001059. |
|
Food and Nutrition Board, Institute of Medicine: Dietary reference intakes for calcium, phosphorus, magnesium, vitamin D, and fluoride. Washington, DC, National Academy Press, 1997. Available at www.nap.edu. |
|
Siega-Riz AM, Herrmann TS, Savitz DA et al: Frequency of eating during pregnancy and its effect on preterm delivery. Am J Epidemiol 153 (7): 647, 2001 |
|
Kaplan M, Eidelman AI, Abolafia V: Fasting and the precipitation of labor. JAMA 250: 1317, 1983 |
|
Knopp RH: Hormone mediated changes in nutrient metabolism in pregnancy: a physiological basis for normal fetal development. Ann NY Acad Sci 817: 251, 1997 |
|
Lebowitz SF: Neurochemical-neuroendocrine systems in the brain controlling macronutrient intake and metabolism. Trends Neurosci 15: 491, 1992 |
|
www.mypyramid.gov/mypyramidmoms/index.html |
|
Moos, MK, et al. Healthier women, healthier reproductive outcomes: recommendations for the routine care of all women of reproductive age. Am J Obstet Gynecol 2008; 199:S280 |
|
Institute of Medicine, Food and Nutrition Board, Committee on Nutritional Status During Pregnancy, part II: Dietary Intake and Nutrient Supplements. National Academy Press 1990; Washington, DC |
|
Hösli I, Zanetti-Daellenbach R, Holzgreve W, Lapaire O. Role of omega 3-fatty acids and multivitamins in gestation. J Perinat Med. 2007;35 Suppl 1:S19-24. Review |
|
Haider BA; Bhutta ZA . Multiple-micronutrient supplementation for women during pregnancy. Cochrane Database Syst Rev. 2006 Oct 18;(4):CD004905. |
|
Goh YI, Bollano E, Einarson TR, et al. Prenatal multivitamin supplementation and rates of congenital anomalies: A metaanalysis. J Obstet Gynaecol Can 2006;28:680–689. |
|
Goh YI, Koren G. Prenatal supplementation with multivitamins and the incidence of pediatric cancers: clinical and methodological considerations. Pediatr Blood Cancer. 2008 Feb;50(2 Suppl):487-9; discussion 498. Review |
|
Kuczkowski KM. Caffeine in pregnancy. Arch Gynecol Obstet. 2009 Feb 24. [Epub ahead of print] |
|
Nehlig A, Debry G. Potential teratogenic and neurodevelopmental consequences of coffee and caffeine exposure: a review on human and animal data.Neurotoxicol Teratol. 1994 Nov-Dec;16(6):531-43. |
|
Maternal caffeine intake during pregnancy and risk of fetal growth restriction: a large prospective observational study. CARE Study Group. BMJ. 2008 Nov 3;337 |
|
Pietrantoni M, Knuppel RA: Alcohol use in pregnancy. Clin Perinatol 18 (1):93,1991 |
|
Chang G, Wilkins-Haug I, Bergman S et al: Alcohol use in pregnancy:improving identification. Obstet Gyaecol 91 (6):892,1998 |
|
Coles C: Critical period for prenatal alcohol exposure. Evidence from animal and human studies. Alcohol Health Res World 18:22,1994] |
|
Pitkin RN: Aspartame ingestion during pregnancy. In Stegink LD, Filer LJ Jr, (eds): Aspartame: Physiology and Biochemistry,pp 555-563. New York, Marcel Dekker,1984 |
|
Sturtevant FM: Use of aspartame during pregnancy. Int J Fertil :30 85,1985 |
|
FAO/WHO Expert Committeee on Food Additives: Toxicological evaluations of certain food additives and food contaminants. Acesulfame potassium. WHO Food Additive Series No. 18 pp 12-27. Geneva, Switzerland, World Health Organization, 1983 |
|
Grice HC, Goldsmith LA: Sucralose: An overview of the toxicity data. Food Chem Tox 38(suppl.2):S1,2000] |
|
Food and Drug Administration: FDA’s approval to sucralose. Federal Register 63:16417, 1998 |
|
Sternfeld B: Physical activity and pregnancy outcome. Review and recoomedations. Sports Med 23(1): 33; 1997] |
|
Artal R, O'Toole M. Guidelines of the American College of Obstetricians and Gynecologists for exercise during pregnancy and the postpartum period. Br J Sports Med. 2003 Feb;37(1):6-12; |
|
Kramer MS, McDonald SW. Aerobic exercise for women during pregnancy. Cochrane Database Syst Rev. 2006 Jul 19;3:CD000180. Review |
|
Ochsenbein-Kölble N, Roos M, Gasser T, Zimmermann R. Cross-sectional study of weight gain and increase in BMI throughout pregnancy. Eur J Obstet Gynecol Reprod Biol. 2007 Feb;130(2):180-6. Epub 2006 May 12 |
|
Outcomes of Maternal Weight Gain, Structured Abstract. May 2008. Agency for Healthcare Research and Quality, Rockville, MD. http://www.ahrq.gov/clinic/tp/admattp.htm |
|
Keppel K, Taffel S: Pregnancy-related weight gain and retention: Implications of the 1990 Institute of Medicine guidelines. Am J Public Health 83: 1100, 1993 |
|
Muscati S, Gray-Donald K, Koski K: Timing of weight gain during pregnancy: promoting fetal growth and minimizing maternal weight gain retention. Int J Obes 20: 526, 1996 |
|
Olson CM, Strawderman MS, Reed RG. Efficacy of an intervention to prevent excessive gestational weight gain. American Journal of Obstetrics and Gynecology 2004;191(2):530–6. |
|
Ludwig DS. The glycemic index: physiological mechanisms relating to obesity, diabetes, and cardiovascular disease. JAMA. 2002;287(18):2414-2423 |
|
Hötzel D. Suboptimal nutritional status in obesity (selected nutrients). Bibl Nutr Dieta. 1986;37(37):36-41. |
|
.[Carmichael S, Abrams B: A critical review of the relationship between gestational weight gain and preterm delivery. Obstet Gynaecol 89;865,1997] |
|
Luke B, Hediger ML, Nugent C, Newman RB, Mauldin JG, Witter FR, O’Sullivan MJ. Body mass index—Specific weight gains associated with optimal birth weights in twin pregnancies. J Reprod Med. 2003;48:217-224. |
|
Luke B, Keith L, Keith D: Maternal nutrition in twin gestations: Weight gain, cravings and aversions, and sources of nutrition advice. Acta Genet Med Gemellol (Roma) 46 (3): 157, 1997 |
|
American Dietetic Association. Position of the American Dietetic Association: Nutrition and lifestyle for a healthy pregnancy outcome. J Am Diet Assoc. 2002;102:1479-1490. |
|
National Academy of Sciences. Nutrition During Pregnancy: Part I: Weight Gain, Part II: Nutrient Supplements. Washington, DC: National Academy Press, 1990. |
|
Scholl TO, Johnson WG. Folic acid: Influence on the outcome of pregnancy. Am J Clin Nutr 2000; 71(suppl 5):S1304-S1307. |
|
Okah FA, Tsang RC, Sierra R, Brady KK, Specker BL. Bone turnover and mineral metabolism in the last trimester of pregnancy: effect of multiple gestation. Obstet Gynecol. 1996;88:168-173. |
|
Story M: Nutritional requirements during adolescence. In McAnarney ER, Kreipe RE, Orr DE et al (eds): Textbook of Adolescent Medicine, pp 75–84. Philadelphia, WB Saunders, 1992 |
|
Scholl TO, Hediger MI, Schall JI et al: Maternal growth during pregnancy and the competition for nutrients. Am J Clin Nutr 60 (2): 183, 1994 |
|
Lenders CM, McElrath TF, Scholl TO. Nutrition in adolescent pregnancy. Curr Opin Pediatr. 2000 Jun;12(3):291-6. Review. |
|
Berenson AB, Wiemann CM, Rowe TF et al: Inadequate weight gain among pregnant adolescents: Risk factors and relationship to infant birth weight. Am J Obstet Gynecol 176: 1220, 1997 |
|
Pencharz PB. Special problems of nutrition in the pregnancy of teenagers. Nestle Nutr Workshop Ser Pediatr Program. 2005;55:213-7; discussion 217-20. Review. |
|
Lenders CM, Hediger ML, Scholl TO, et al: Gestational age and infant size at birth are associated with dietary sugar intake among pregnant adolescents. J Nutr 1997;127:1113–1117. |
|
Scholl TO, Hediger ML, Khoos CS, et al: Maternal weight gain, diet and infant birth weight: Correlations during adolescent pregnancy. J Clin Epidemiol 1991;44:423–428. |
|
Wallace HM, Weeks J, Medina A: Services for pregnant teenagers in the large cities of the United States, 1970–1980. JAMA 1982;248:2270–2273. |
|
Dubois S, Coulombe C, Pencharz PB, et al: Nutrition intervention during adolescent pregnancy. J Am Diet Assoc 1997;97:871–878. |
|
Formanek R: Focuses on the food safety rules issued by the United States Food and Safety Administration Center for Food Safety and Applied Nutrition in January 2001. How to reduce the risks for Listeria monocytogenes bacterium; Health risks of pregnant women and women considering pregnancy regarding fish with mercury; Safety of imported food and eggs. FDA Consumer 35 (2): 15, 2001 |